- Markets & Solutions
- Products
- Capabilities
- Industrial Solutions
- Industrial Solutions Overview
- High Temperature Silicone Seals and Silicone Gaskets
- Silicone Rubber Tubing
- Architectural Building Gaskets
- Silicone Moulding
- Dielectric Silicone Sleeving in Electricity
- Silicone Sponge and Silicone Foam
- Silicone Prototype and Design
- Our Commitment to Quality
- Literature and Downloads
- Quality
- Markets & Solutions
- Products
- Capabilities
- Industrial Solutions
- Industrial Silicone Solutions Overview
- High Temperature Silicone Seals and Silicone Gaskets
- Silicone Rubber Tubing
- Architectural Building Gaskets
- Silicone Moulding
- Dielectric Silicone Sleeving in Electricity
- Silicone Sponge and Silicone Foam
- Silicone Prototype and Design
- Our Commitment to Quality
- Literature and Downloads
- Quality
- Markets & Solutions
- Products
- Capabilities
- Industrial Solutions
- Industrial Silicone Solutions Overview
- High Temperature Silicone Seals and Silicone Gaskets
- Silicone Rubber Tubing
- Architectural Building Gaskets
- Silicone Moulding
- Dielectric Silicone Sleeving in Electricity
- Silicone Sponge and Silicone Foam
- Silicone Prototype and Design
- Our Commitment to Quality
- Literature and Downloads
- Quality
- Markets & Solutions
- Products
- Capabilities
- Industrial Solutions
- Industrial Silicone Solutions Overview
- High Temperature Silicone Seals and Silicone Gaskets
- Silicone Rubber Tubing
- Architectural Building Gaskets
- Silicone Moulding
- Dielectric Silicone Sleeving in Electricity
- Silicone Sponge and Silicone Foam
- Silicone Prototype and Design
- Our Commitment to Quality
- Literature and Downloads
- Quality
- Markets & Solutions
- Products
- Capabilities
- Industrial Solutions
- Industrial Silicone Solutions Overview
- High Temperature Silicone Seals and Silicone Gaskets
- Silicone Rubber Tubing
- Architectural Building Gaskets
- Silicone Moulding
- Dielectric Silicone Sleeving in Electricity
- Silicone Sponge and Silicone Foam
- Silicone Prototype and Design
- Our Commitment to Quality
- Literature and Downloads
- Quality

Markets & Solutions
 Medical
Medical
Top medical device companies from around the world come to Silicone Altimex for high quality, high performance components like valves, seals and Altimed® tubing.
Operating Class 7 Cleanrooms under ISO 13485 and ISO 14644 quality and environmental management systems, the company has the capabilities for manufacturing FDA-registered products that meet extremely stringent standards.
From material development to manufacturing to assembly, you can count on the experts at Silicone Altimex to deliver excellent custom products requiring tight tolerance and precision.
Bring us your project needs, we will exceed them.
Customers prefer our solutions for:
- Feeding tubes
- Infusion pump tubing
- Cardiovascular pump tubing
- Valves
- Seals
Pharmaceutical
 At Silicone Altimex, we know that bringing a pharmaceutical drug or drug delivery device to market is a long, expensive process, and we work hard to make it easier for our customers. From large pharmaceutical companies to new start-ups and everything in between, we can help streamline the process.
At Silicone Altimex, we know that bringing a pharmaceutical drug or drug delivery device to market is a long, expensive process, and we work hard to make it easier for our customers. From large pharmaceutical companies to new start-ups and everything in between, we can help streamline the process.
Silicone Altimex’ material development and engineering teams are dedicated to helping you make your testing and approval process seamless, less cumbersome, quicker and more cost-effective. We offer both standardized and custom-made products, each crafted with the same dedication and attention to detail that has made Silicone Altimex a leader in the pharmaceutical industry. Our team will help make you more agile and responsive to the market without sacrificing quality or reliability.
Specializing in custom bio-pharma transfer set production, the Silicone Altimex team will assist with the design and development of any system from concept to finished sterilized product.
Audited and approved by blue-chip pharmaceutical companies, Silicone Altimex stands ready to provide:
- Single use bio-pharmaceutical assemblies
- Pharmaceutical synthesis rigs
- Bio filtration silicone assemblies
- Sterile fittings
- Braided silicone tubing
- Electronic termination plugs
- Customer specific packaged kits
Learn more
Ask a question, request a quote or learn more about Silicone Altimex and Silicone Altimex career opportunities:
ContactRequest a QuoteCareersAbout


Navigate
Silicone Altimex
Contact Us
Global Headquarters
Silicone Altimex
Manufacturing and Engineering Facility
49 Pasture Road
Stapleford, Nottingham, NG9 8HR
0115 949 6890
Additional ways to contact us
Silicone Altimex & Q Holding Locations
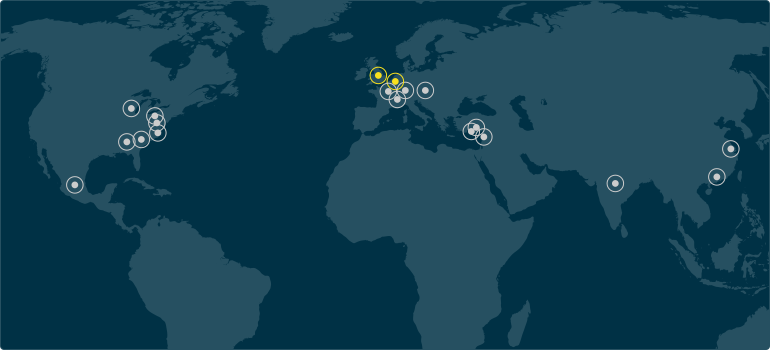 View all Silicone Altimex location details.
View all Silicone Altimex location details. Silicone Altimex Location
Additional Q Holding Company Location
©2024 Silicone Altimex, a SaniSure Company • All Rights Reserved • Terms & Conditions
Track Your Silicone Altimex Shipment
For order tracking in Jasper, GA, contact your QSR customer service representative:
Vikki Stanger
vstanger@qsr-inc.com
706-692-8626
For order tracking in China, contact your QSR customer service representative:
Mary Zheng
mary@qsrchina.com
+ 86-769-836-959-90
For order tracking in Jasper, GA, contact your QSR customer service representative:
Vikki Stanger
vstanger@qsr-inc.com
706-692-8626
For order tracking in China, contact your QSR customer service representative:
Mary Zheng
mary@qsrchina.com
+ 86-769-836-959-90